Hydrogen Peroxide is a common additive in hydroponics and can be used to prevent disease, as an oxygenator, and as a disinfectant / cleaner. It is a very potent additive and should be handled carefully. Here we will go through the pros and cons of using hydrogen peroxide while growing.
What is hydrogen peroxide?
Hydrogen Peroxide or H2O2 has been a common additive used in hydroponics for several decades. It is simply water with an extra oxygen atom attached. While this may sound innocent, H2O2 is very powerful and comes with many risks.
⠀⠀⠀⠀⠀⠀
On its own water, or H2O, is a very stable molecule, but sticking an extra oxygen onto it makes for instability. This means it is very reactive and can spontaneously decompose into water and oxygen.
⠀
How does hydrogen peroxide control pathogens?
Plants and animals produce hydrogen peroxide under their normal metabolic functions. Since it is potentially highly toxic and reactive, plants and animals are well protected against it by having enzymes known as peroxidases that can break it down quite quickly. On the other hand, many types of microbes don’t have this ability and this gives us an advantage to exploit! Low concentrations of hydrogen peroxide can be harmful to microbes, but since plants can break it down, they’re immune. At a 29% concentration, plants can be fed a 1-2 mL per liter nutrient solution of hydrogen peroxide.
⠀
⠀
Warning: never use hydrogen peroxide with beneficial microbes. It will kill all the good bacteria along with the harmful bacteria!
⠀
⠀
Does hydrogen peroxide work as a disinfectant?
Yes! Along with feeding directly to plants with nutrients, hydrogen peroxide can be used as a general disinfectant around your grow room. It can disinfect pots, floors, and other equipment. Since it breaks down into water and oxygen, it won’t leave any residue. To use it as a disinfectant, dilute with 20 parts water to 1 part hydrogen peroxide along with a drop of detergent to help it penetrate. Also keep in mind, disinfectants work best once a surface is already clean so it is not losing potency by reacting with dirt and grime. Always use protective gloves when handling concentrated hydrogen peroxide.
⠀
How to oxygenate soil using hydrogen peroxide:
If you have ever pulled up a potted plant out of its container along with the soil you will notice most of the roots wind up clustered around the drain holes. It is commonly known that plants release more oxygen than they absorb, however, this is only the case for the leaves during photosynthesis! Roots have to take their oxygen from their environment, but this means in dense compacted soil there is little oxygen being circulated which can result in poorly developed root systems. The roots will seek out any potential oxygen source which is why they cluster around the drain holes.
Since there is a lot of material in the soil for hydrogen peroxide to react with, it will quickly break down and liberate the oxygen. This is an excellent way of quickly and evenly distributing oxygen throughout all of the soil.
⠀
⠀
⠀
⠀
Are there any safety concerns with hydrogen peroxide?
Yes, one should always be careful when handling any chemicals but especially hydrogen peroxide. In higher concentrations, hydrogen peroxide can burn skin and is very dangerous to your eyes. Because it can break down oxygen, if in a sealed container it could build up pressure and explode. The hotter the temperature the more rapidly oxygen is liberated and the more cause there is for concern. Light can also poses issues to hydrogen peroxide so it is better stored in a cool dark place. As with any chemical product, it is recommended to consult the safety data sheet (SDS) for detailed information on safe handling!
⠀⠀
Under no circumstances should Hydrogen Peroxide be ingested orally.
⠀
Always be careful when using hydrogen peroxide and never ingest it. There are many claims of Hydrogen Peroxide being a miracle cure, but the scientific evidence to back this up is questionable. Under no circumstances should Hydrogen Peroxide be ingested orally. It will breakdown inside of the stomach and form oxygen which can increase the pressure exponentially to the point of ruptures possibly occurring.
⠀
⠀
How long does hydrogen peroxide stay good for?
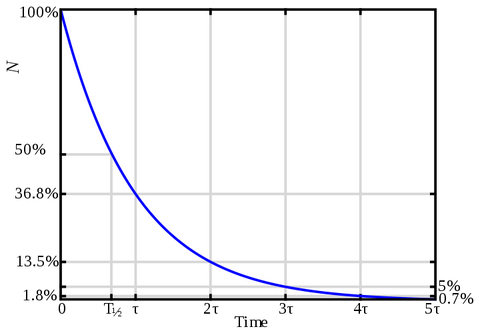
As previously stated, hydrogen peroxide is a very unstable molecule and will degrade over time. This depends on many factors: concentration, light, pH, and most importantly the amount of impurities there are to react with. For example, soil contains many things for hydrogen peroxide to react with and so it won’t stay active for long. Furthermore, its concentration in a nutrient solution will decrease by 50% effectiveness over a set amount of time and from there will again decrease by another 50% over the same amount of time, essentially leaving it at zero.
⠀⠀⠀⠀⠀⠀
The half life of everyone’s solution is going to be different. For example, hydrogen peroxide that is kept cool and clean will keep the longest versus if the hydrogen peroxide was in a nutrient solution with high levels of organic material, it would have a short life. Unfortunately there is no way for the average user to be able to calculate the life of their hydrogen peroxide, but it is important the grower is aware the potency can go down very quickly. By the time the next feeding comes along there might not be any hydrogen peroxide remaining!
⠀
⠀
⠀
⠀
Bonus Use:
While unrelated to growing, this may be of interest to some readers. It has become trendy to play and collect video games and personal computers from the 80s and 90s, but a lot of the time the plastic has discolored into an ugly yellow-brown color. An application of hydrogen peroxide under ultraviolet light can eliminate the discoloration and restore the plastic to its original color! There are several do it yourself recipes and methods available online.
⠀⠀⠀⠀⠀
⠀⠀
⠀⠀⠀⠀⠀
Retrobright, or Retr0bright, is a hydrogen peroxide mixture used to clean old game cartridges and computers!
Check out our Vinny's Tip on Growing with H₂O₂ HERE!
Any questions? Email us at marketing@futureharvest.com or send us a message on Instagram @futureharvestdevelopment and we will help you as best we can!





4 comments
Hi William,
Yes H202 will eliminate the green mold on the outside of your grow bag.
Thanks for reaching out, and have a great day,
Will hydrogen peroxide eliminate green mold starting on the outside of a fiber grow bag
Keeping in mind that our product is not registered as a pesticide any advice given is generic and is not meant specifically for the Retrobrite product that we sell.
It looks as though an ORP level of 750-790 is adequate to kill zoospores within 5 minutes. That would be in laboratory conditions however, in the real world there are more complications such as the zoospores being able to “hide” in the cracks and crevices of the plastics in your growing system ect. You will never be able to completely kill it off, the best that you can hope for is the slow it down enough to keep your plants healthy until harvest. What is the temperature of the nutrient solution? Lower temperatures will also help to slow it down.
Ideally when your harvest is finished you will want to dispose of the equipment that has come in contact with the Pythium as it will be contaminated which can cause it to show up in future crops. If that isn’t an option, circulate a strong bleach solution through the system for 24 hours. Follow by water to get rid of any bleach contaminants.
Let us know what ORP levels are required to eliminate Root rot and how to do.
I am thinking bout using 3% dilution at three mil per.
Res change has seen the ORP level as high as 500MV due to tap water and maintained at 350MV.
In the middle (week 7) for 10-week fruit of using FH full program for hydro in ebb n flow.
Got meters for ORP levels,
Two male plants were removed from the table with the ass-end still there to be where nasty is.
All in all, looking forward to harvest